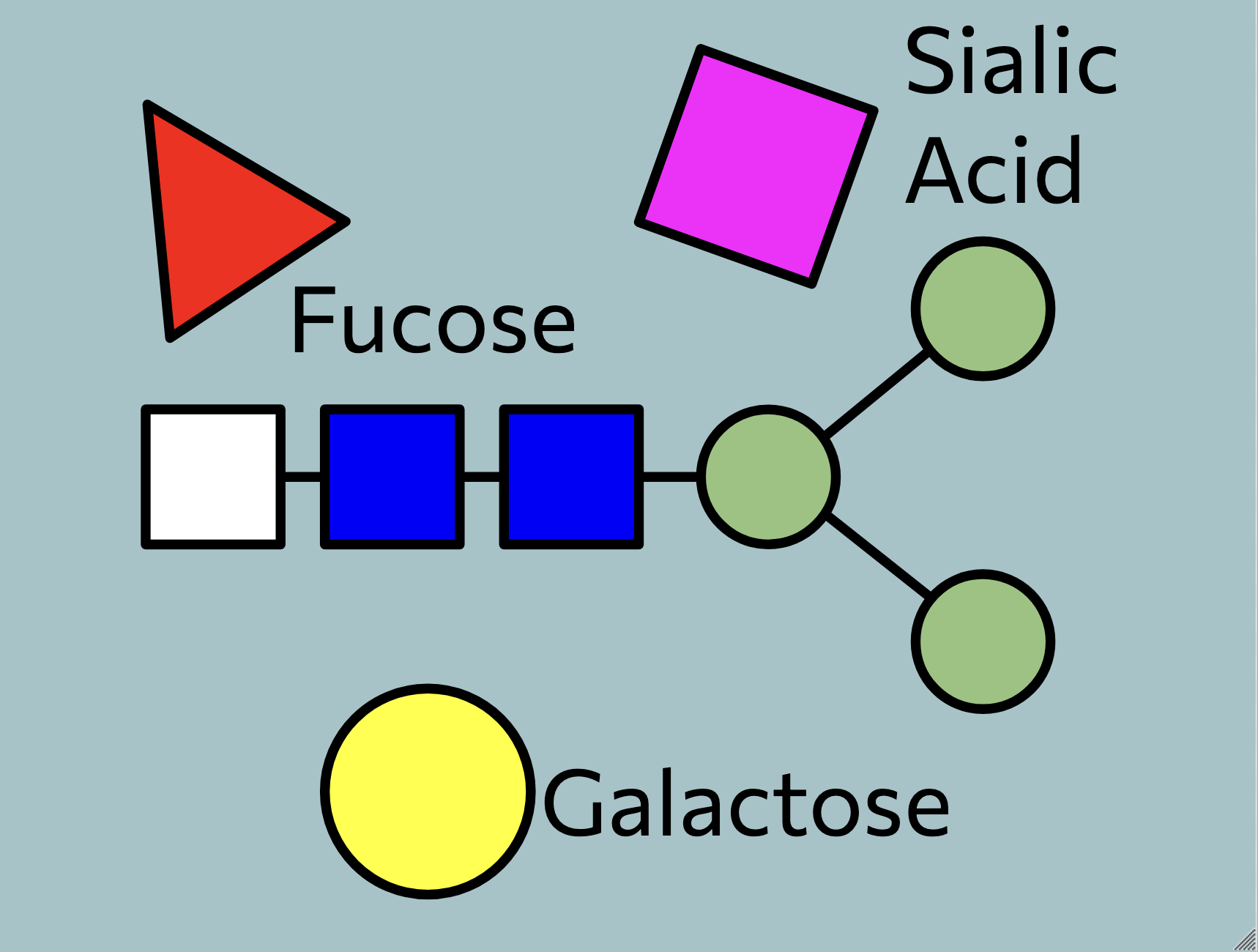
N-Linked Glycosylation
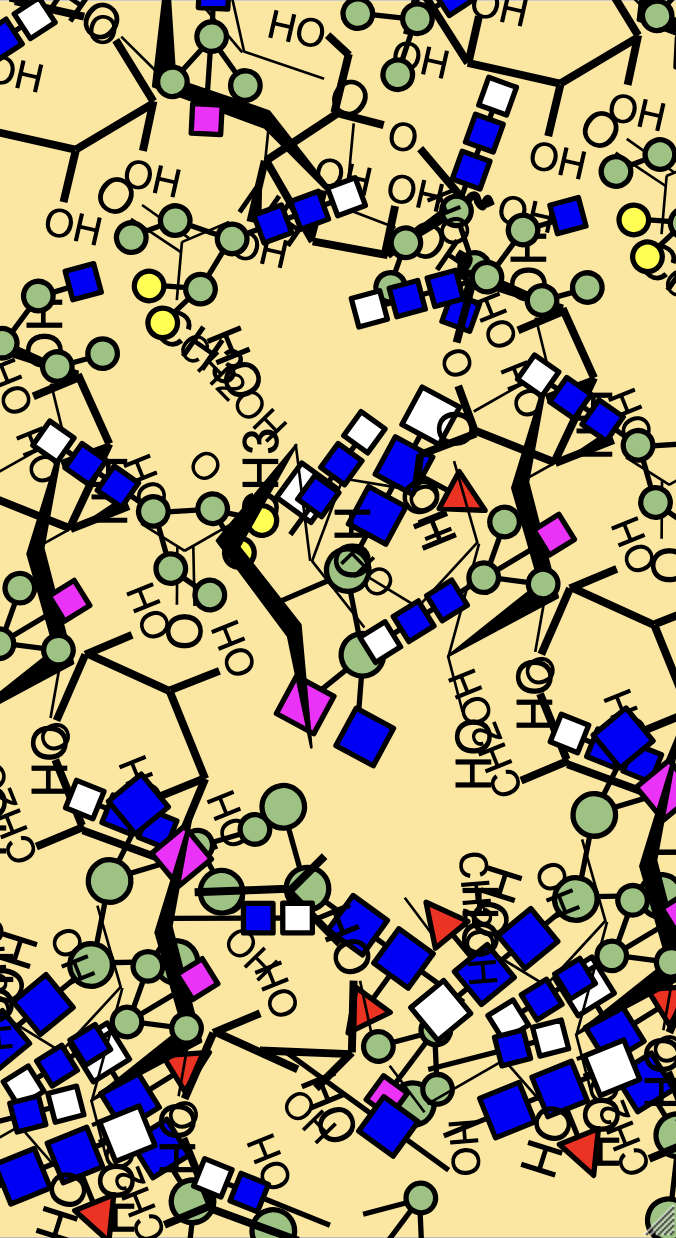
High-Mannose Class
Studies and Research on N-Glycans



Within the eukaryotic cell, proteins that are made must be checked for degradation and have their general quality examined. N-glycans are protein-attached biological markers that other chaperone proteins and enzymes can read to determine the quality of a given protein. N-glycans differ widely between different types of proteins, cells, and organisms, but they all have general classifications and structures (UniProt, 2019). Through studying this special class of molecules, many researchers have found promising therapies and treatments to disorders that manifest in faulty protein manufacturing within the cell.
N-glycans start their creation in the rough endoplasmic reticulum (ER) and go through the following formation process:
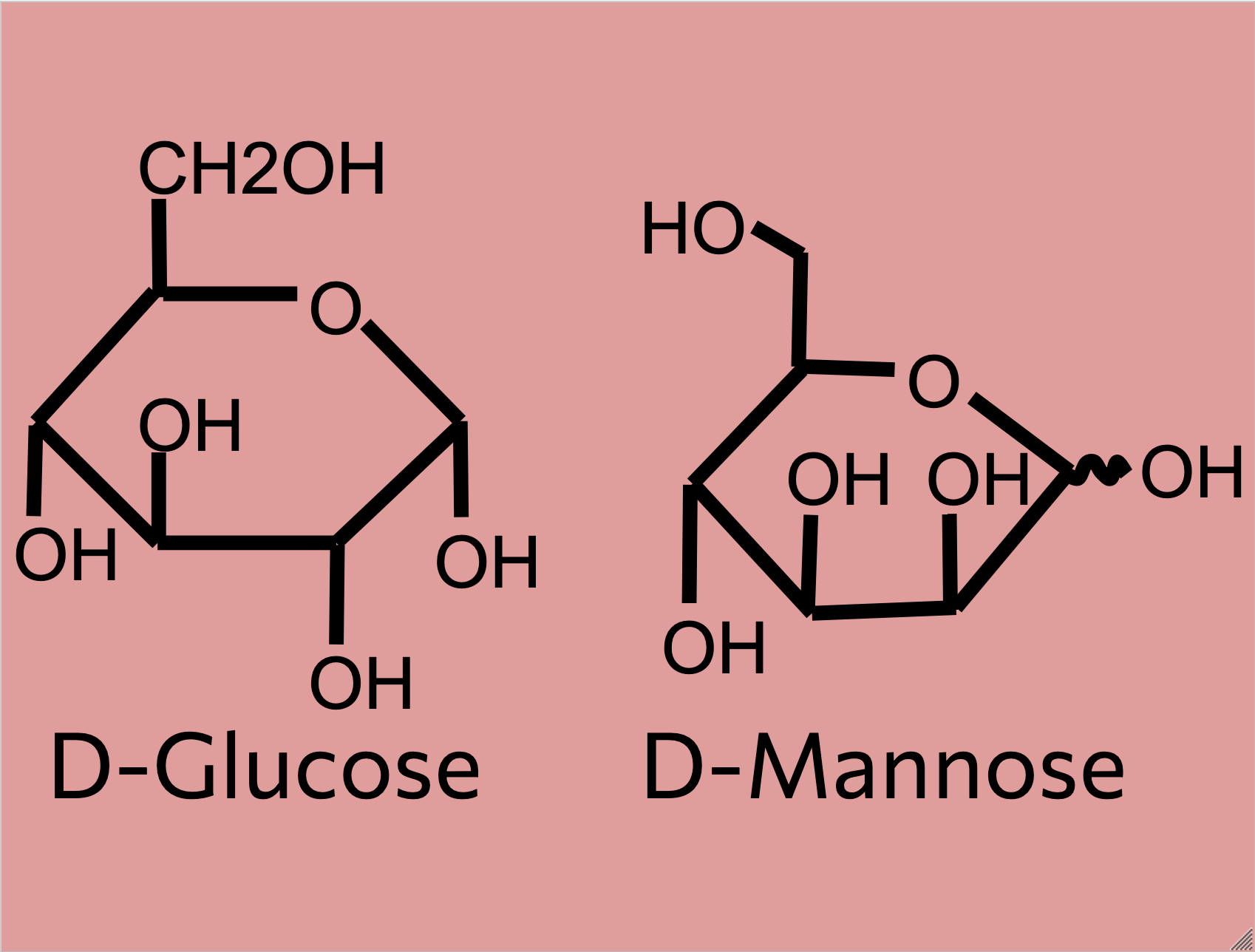
Within the rough ER, blocks of 14 sugars are added to the nascent polypeptide chain of the n-glycan and 3 glucose with 1 mannose residue are cleaved off before the N-glycan is released to the golgi body. Within the golgi body, the N-glycan undergoes a complex “pruning” process where branches are removed and/or added to it (UniProt, 2019). This allows for the N-glycan to be differentiated into one of three classes (high-mannose, hybrid, complex). The structure of N-glycans is part of the larger topic of glycan/carbohydrate synthesis in the body, which is studied by glycobiologists. Additionally, different organisms will have different N-glycan structures corresponding to the organelles within that organisms’ cells (UniProt, 2019). The N-glycans tell proteins at the end of the golgi body’s signal pathway if the protein the N-glycan is attached to was made correctly or not. Once the protein and n-glycan are done, they are packaged in a vesicle to be transported to other areas in the cell or to places outside of the cell.
The three main classes of N-glycans are explained further in their corresponding sections:
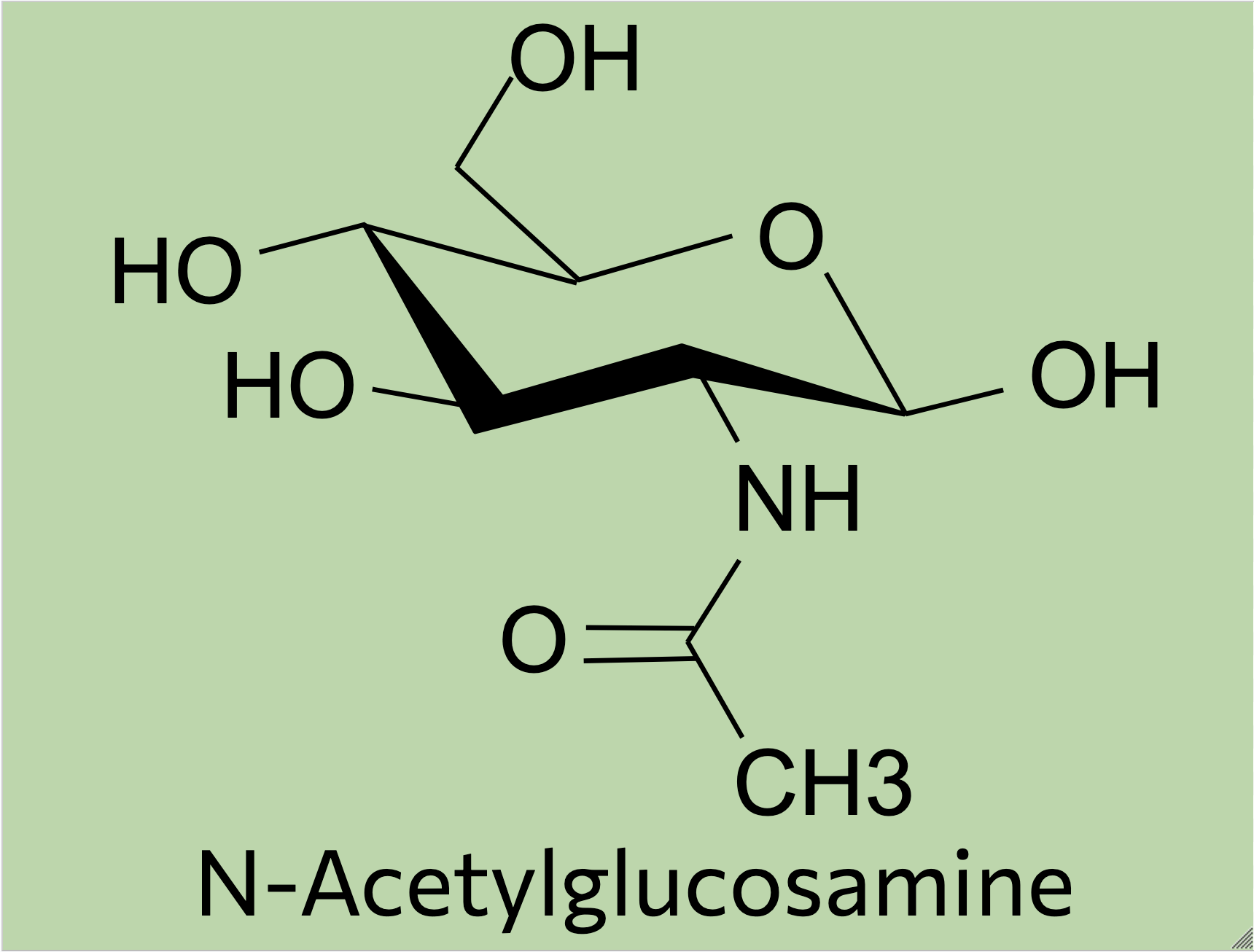
Generally, N-glycans start off as a simple mannose chain before having additional branches to become a high-mannose N-glycan, substitutions to become a hybrid N-glycan, or the addition of GlcNAc (N-acetylglucosamine) residues and other molecules to become a complex N-glycan (Sigma Aldrich, 2024).
N-glycans are modified further when moving through the golgi body through various things such as the addition of amino acids to form a glycoprotein. Certain parts of the pathway focus on breaking down N-glycans and recycling them through reglycosylation in order to correct errors in the N-glycans’ structures (Sigma Aldrich, 2024). Enzyme deficiencies in the pathways that create N-glycans can lead to metabolic disorders due to the build-up of misfolded proteins and old N-glycans that are never broken down.
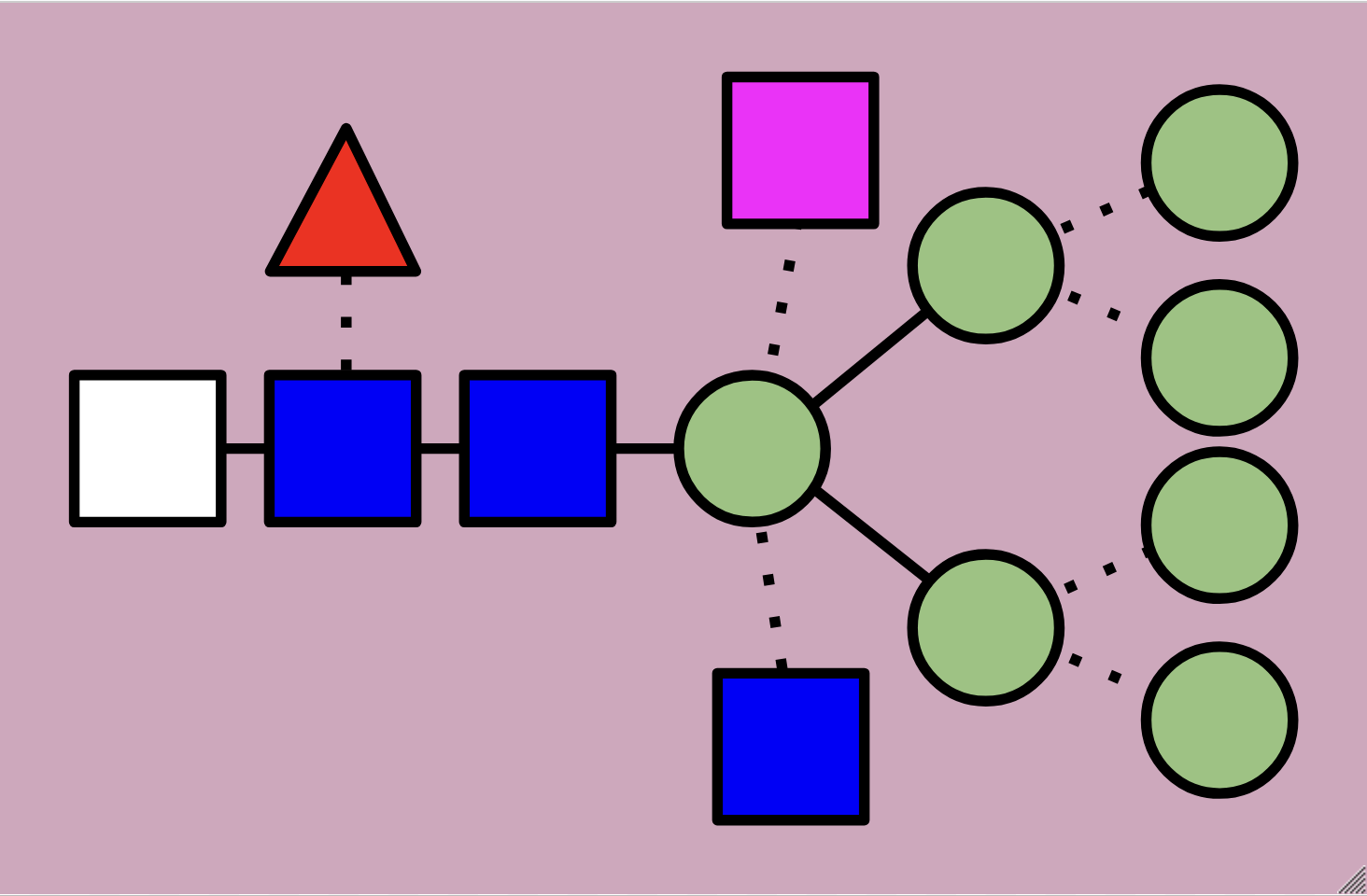
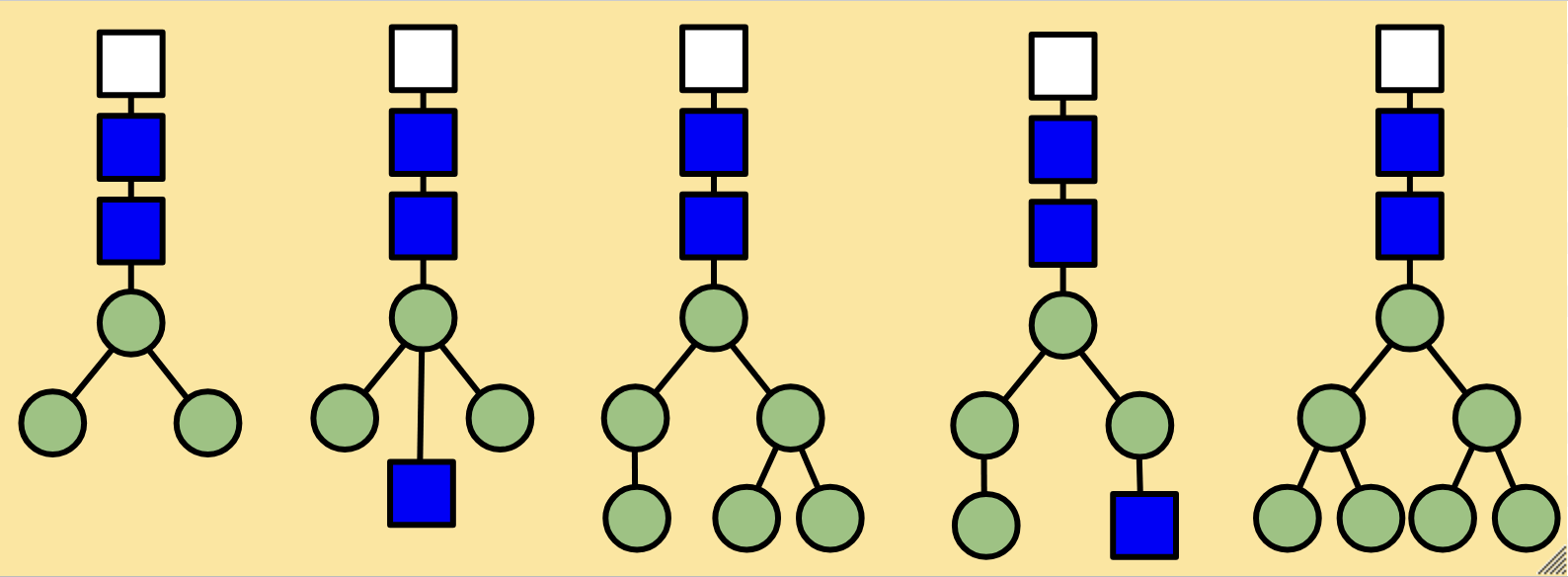
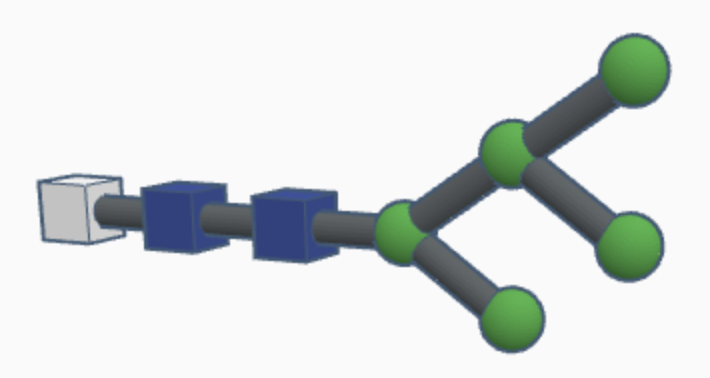
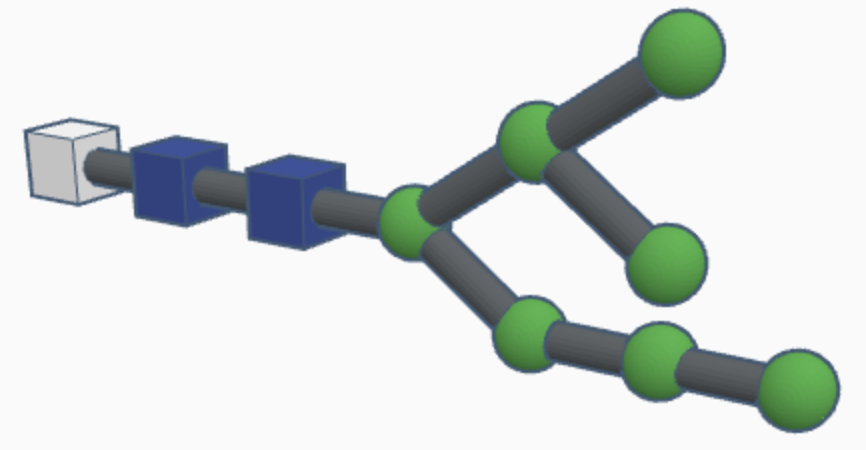
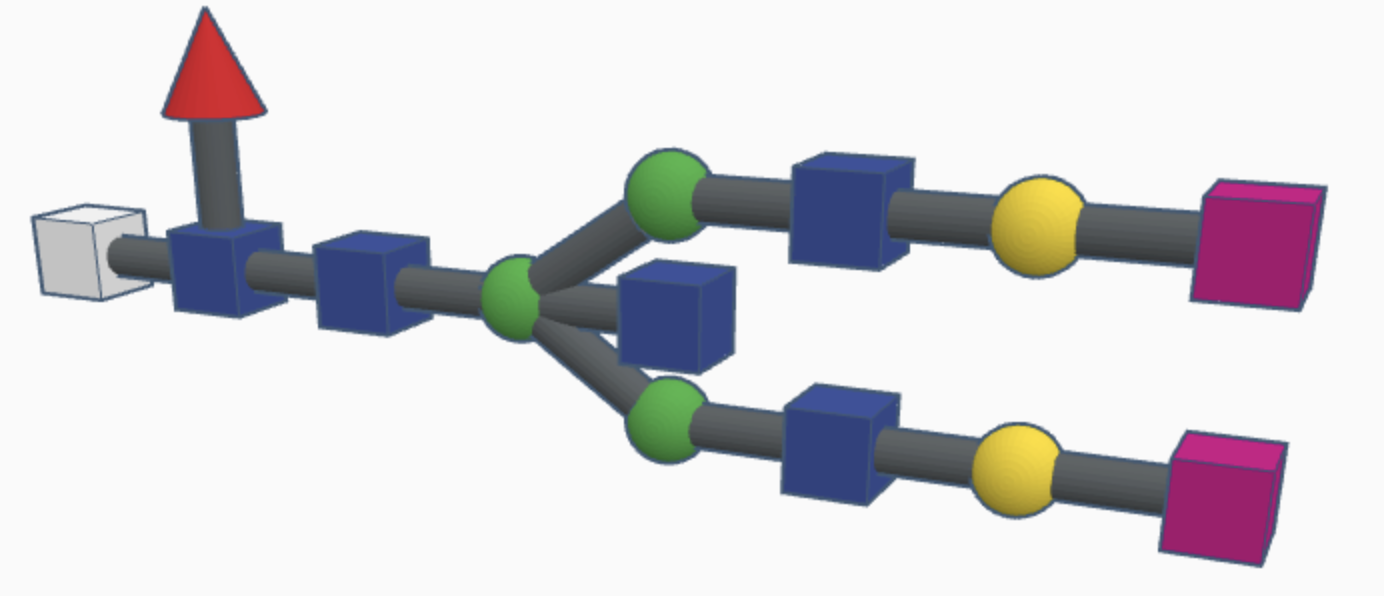
High-mannose N-glycans have a core N-acetylglucosamine (GlcNAc) chain (with beta connections) connected to the nitrogen atom on one end and connected to unsubstituted terminal mannose sugars on the other. Most N-glycans start out in a low mannose structure with only two mannose sugars branching off from the GlcNAc chain, their connections named alpha-3 and alpha-6. To form a high-mannose N-glycan, many branching alpha connections are made with more mannose sugars, creating a complex pattern that can be read by chaperone proteins.
Examples of high-mannose N-glycans are shown below.
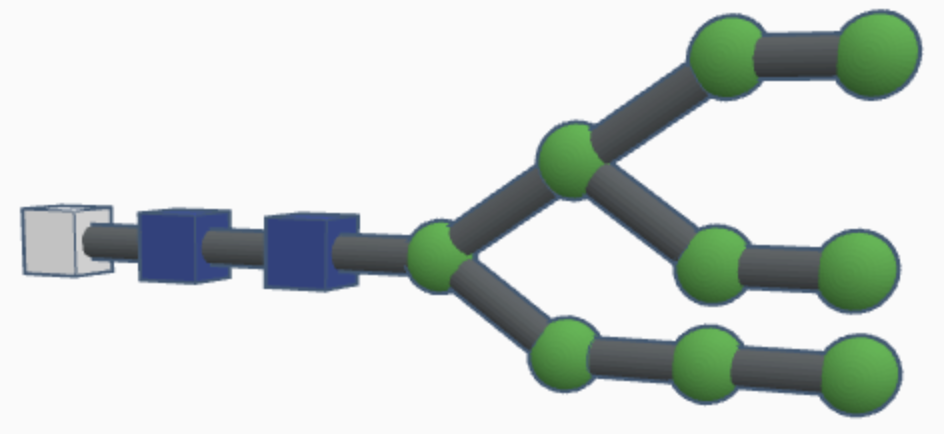
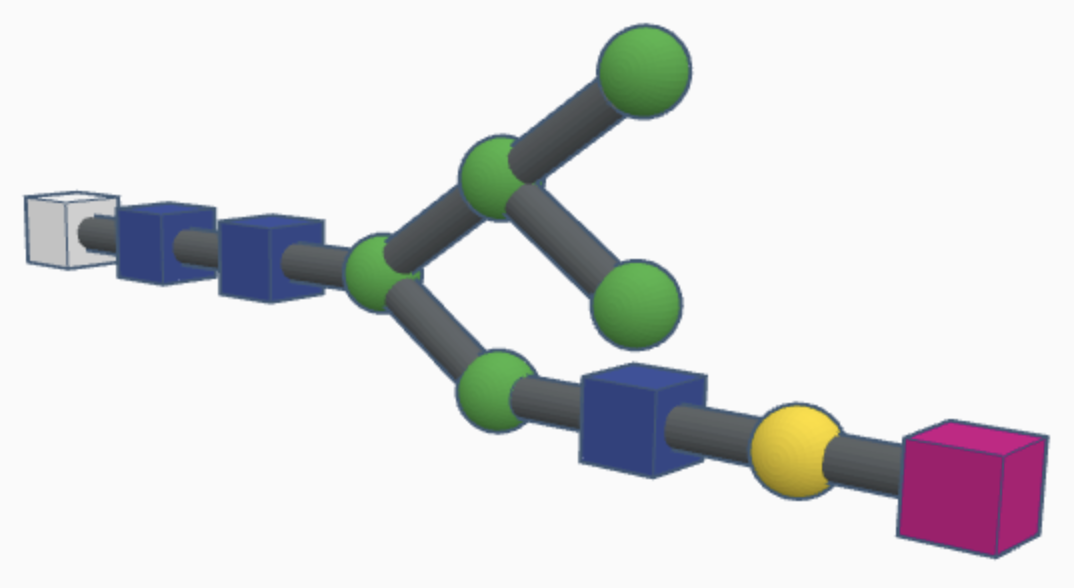
While hybrid N-glycans have a mixture of substituted and unsubstituted mannose sugars, complex N-glycans only have substituted mannose sugars branching off of the main mannose chain. This means that the alpha-3 and alpha-6 mannose sites are the only unsubstituted mannose sugars in the N-glycan, with GlcNAc branching off of them to form branching chains of substituted mannose sugars. Additionally, fucosylation causes a fucose sugar to be added near the start of the GlcNAc chain for some complex N-glycans. The high variety of sugars causes complex N-glycans to be harder to break down during the reglycosylation process within the signal pathway, which means that they’re more durable but it’s harder to correct mistakes in their structure as a result.
Examples of hybrid N-glycans are shown below.
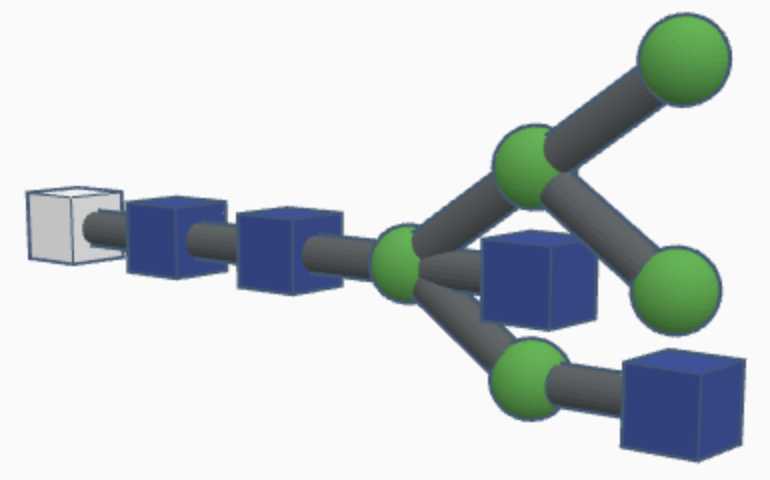
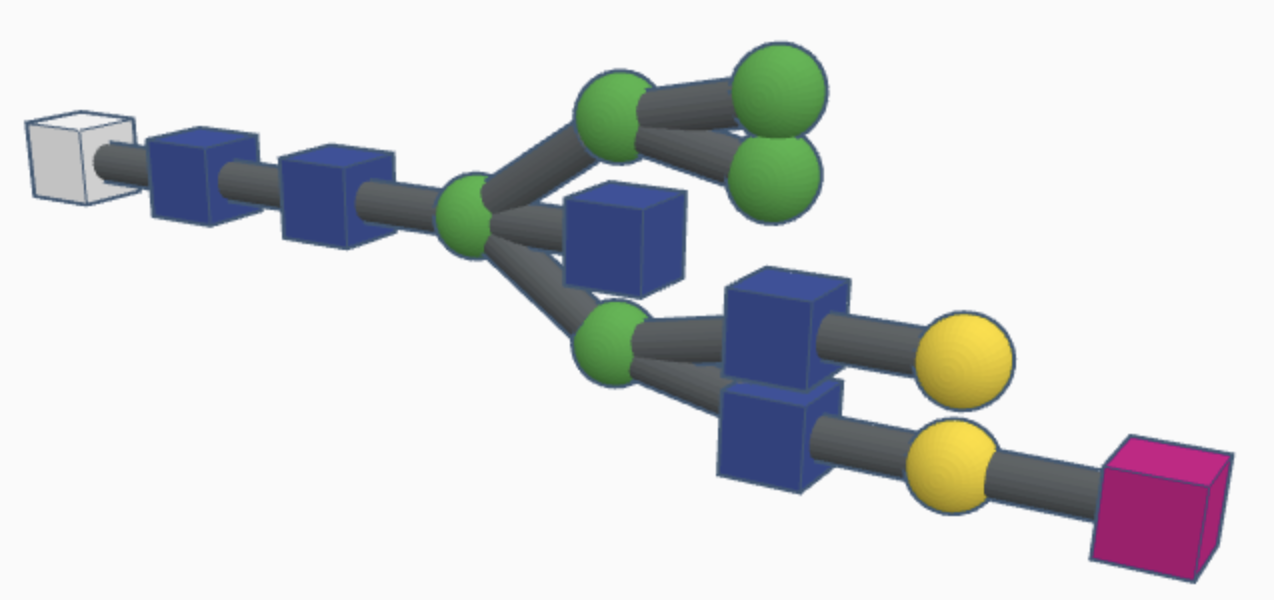

Hybrid N-glycans are similar to high-mannose N-glycans but with additional carbohydrates attached to the branching paths of mannose sugars (called substituted mannose sugars). This continued differentiation allows for more types of N-glycans to be made and stabilizes the branches with more rigid molecules than just mannose sugars.
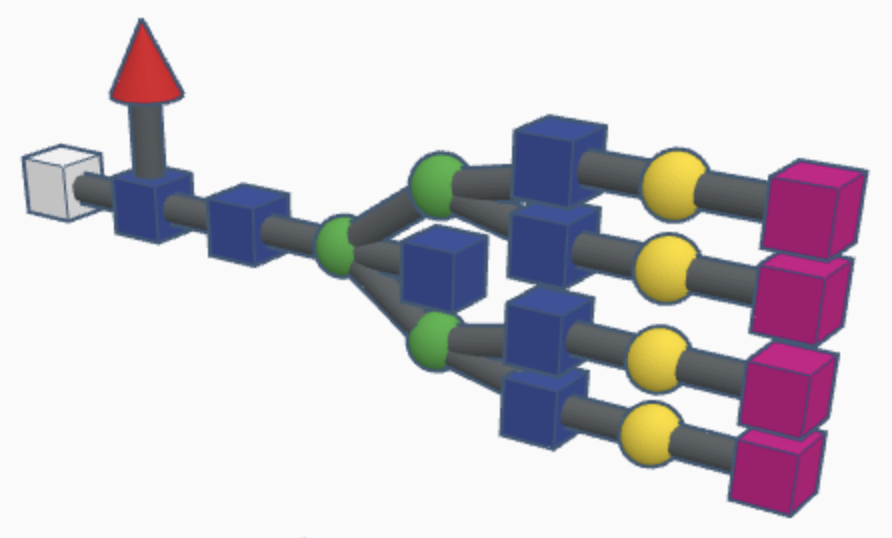
Examples of complex N-glycans are shown below.

Many of the following studies have researchers looking into a specific biological pathway or metabolic system, and this is because major protein disorders often manifest from chaperone proteins being unable to read N-glycans correctly to assess a protein's quality. This allows for misfolded proteins to accumulate and damage cells, which can have serious consequences if the disorder is widespread enough. By studying N-glycan synthesis, researchers can create promising new therapies to mitigate disorders on a cellular level, allowing for targeted treatments. Thus, glycobiology is a widely studied field of molecular biology due to its useful applications for protein disorders.
Alpha-1 antitrypsin (AAT) and antithrombin III (ATIII) are two human serpins, serine protease inhibitors, which have a complex glyco-code that dictates the structure of the n-glycans attached to them in the endoplasmic reticulum signal sequence. AAT has glycans near its N terminus (start of the protein) and ATIII has glycans near its C terminus (end of the protein) but they both undergo similar processes that end up leading to reglycosylation by the UGGT (UDP-Glucose Glycoprotein Glucosyltransferase) enzyme. This study looked at different cases when AAT and ATIII were folded correctly or incorrectly in order to judge the efficiency of certain glycans. They also looked at mutations that may have affected the creation of n-glycans or the reglucosylation done by UGGT at the end of the signal pathway (Guay et. al, 2023).
Immunoglobulin G (igG) is an important antibody of the immune system, and its production is changed depending on the specific undergoing immune response. In this study, n-glycans that change the functions of igG are examined, along with many other molecular attachments. These molecules can cause the secretion of cytokines that affect inflammation, so n-glycans play a large role in immune function by affecting the immune proteins that create the signals to attack certain infections. Different classes of glycoprotein are examined, along with their effects on different immune cells and immune responses. Potential neurologic treatments that can be developed from igG protein engineering are discussed near the end of the article (Kronimus et. al, 2019).
By studying diseases of the n-glycosylation pathway, researchers in this study found that O-GlcNAcylation changes the expression of two rate-limiting proteins in the pathway, changing the speed that n-linked glycosylation takes place. Researchers found that in the inherited disorder PMM2-CDG, UDP-GlcNAc and O-GlcNAcylation were present in higher levels, which increased O-GlcNAcylation and thus affected the pathway. Two different glycosylation pathways were studied, one normal and one mutated, and the researchers found that mutations that caused increases in certain proteins on the pathway could protect from the impact of this disorder. Zebrafish cells were used in this study, and the pmm2^m/m mutation was applied in the second pathway (Matheny-Rabun et. al, 2024).